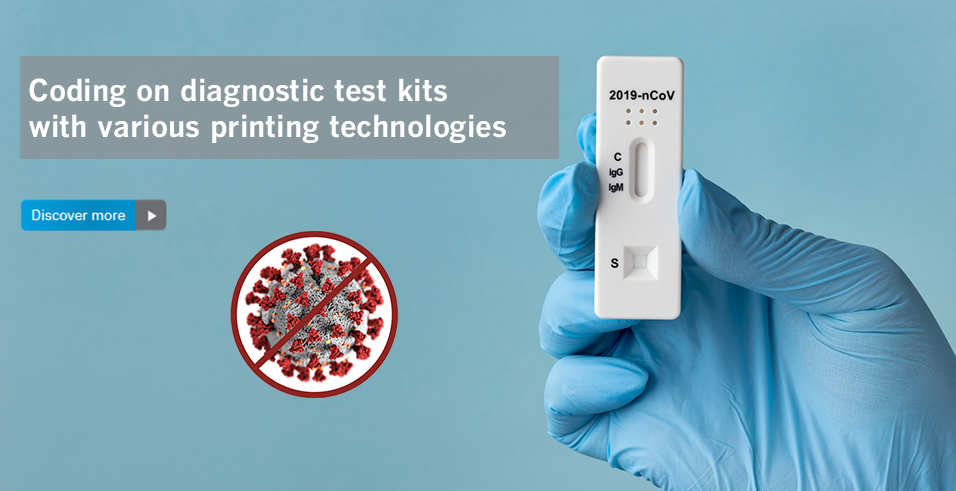
Marking & Coding Solutions for Pharmaceutical & Medical Device Products
Latest News
Equipment Sales: +971 50 127 8532
Products and Services

Product recommendations
Continuous Inkjet, Thermal Inkjet, Laser Marking Systems, Thermal Transfer Printers, Large Character Marking, Label Print & Apply
Which coding solution for my substrate?
Gain an understanding into which coding solution best fits your pharmaceutical and medical device packaging

Code Quality Test Service IQ/OQ Documentation
Discover the Videojet Pharma Line Code2Carton service for superior code quality and our IQ/OQ validation packages.
Case Studies

Case Studies
Understand how our products and services have enhanced our customer’s pharmaceutical and medical device production lines.
Coding Regulations

Pharmaceuticals
This section gives an overview of the global coding requirements related to serialization regulations.
Medical Devices
Find out the coding requirements for medical device manufacturers that derive from the U.S. UDI system and the European Medical Device Regulation